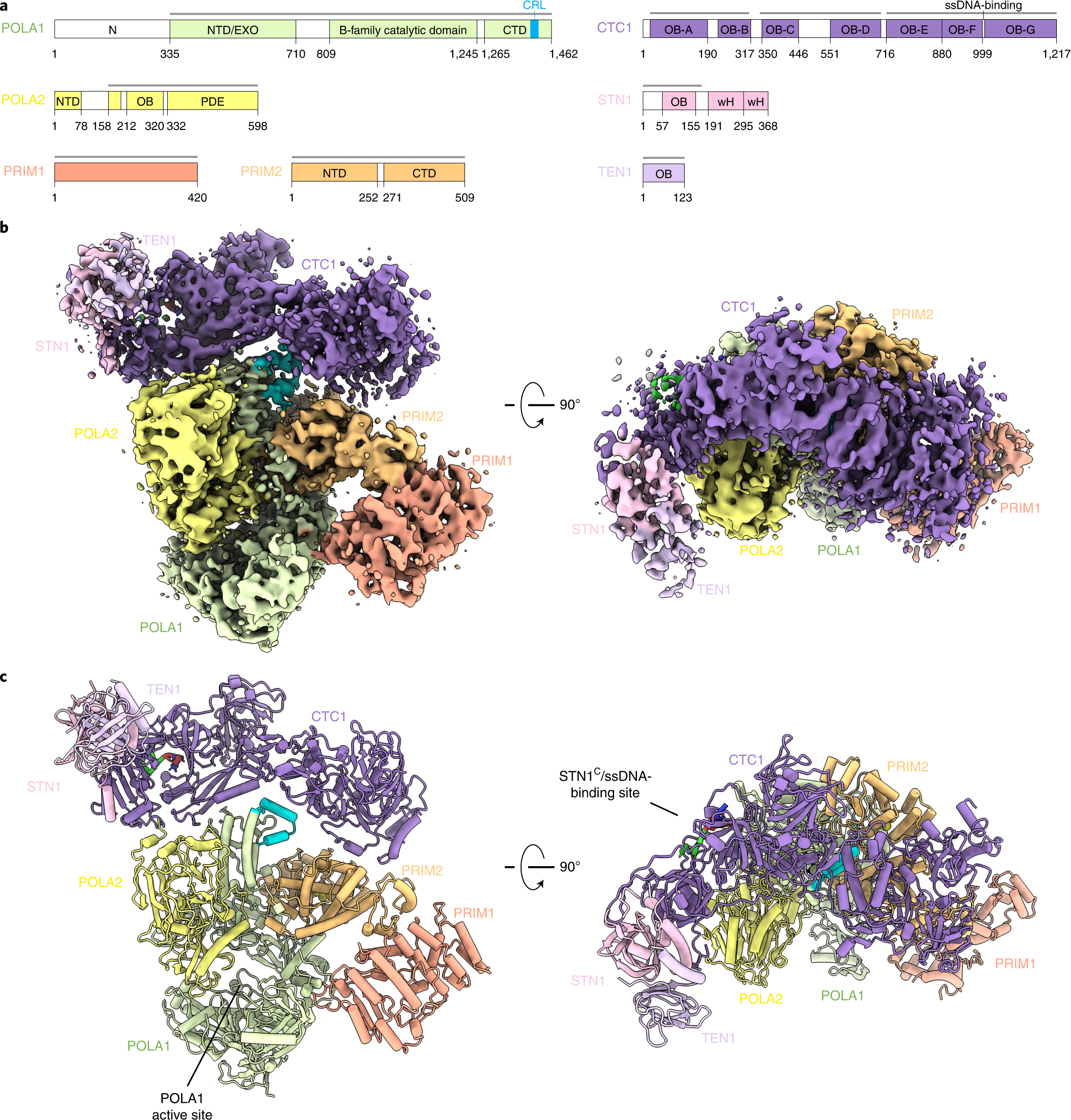
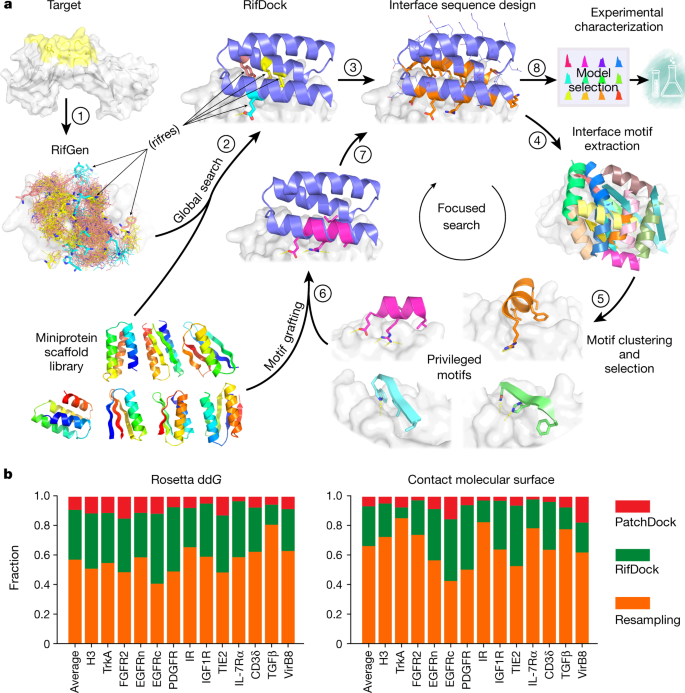
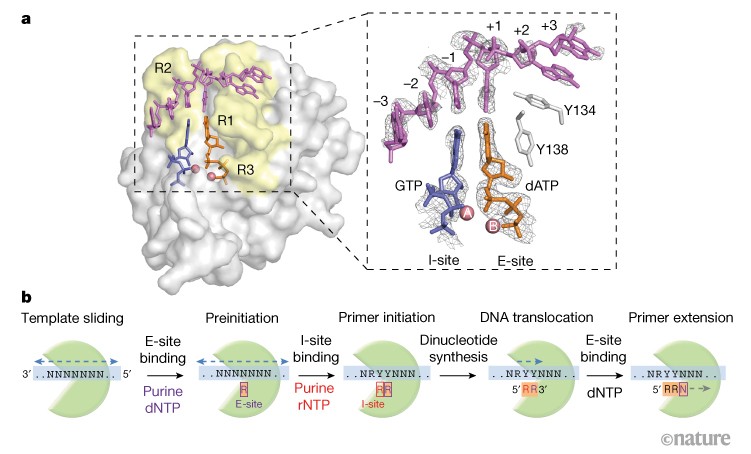
The selection process of licensing a DNA mismatch for repair | Nature Structural & Molecular Biology

Structure of the metastatic factor P-Rex1 reveals a two-layered autoinhibitory mechanism | Nature Structural & Molecular Biology
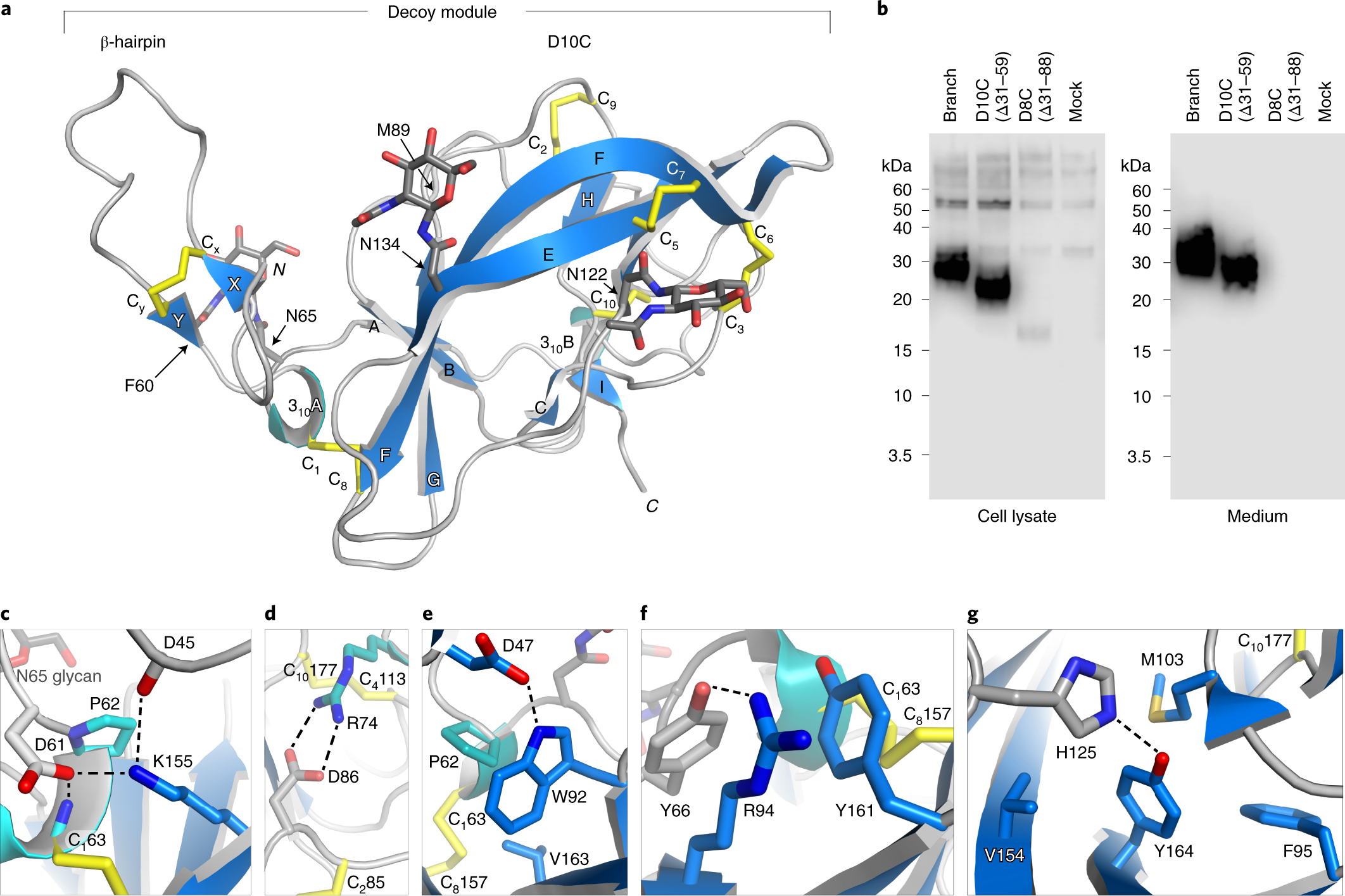
Structure of the decoy module of human glycoprotein 2 and uromodulin and its interaction with bacterial adhesin FimH | Nature Structural & Molecular Biology

Synergistic activation of the insulin receptor via two distinct sites | Nature Structural & Molecular Biology
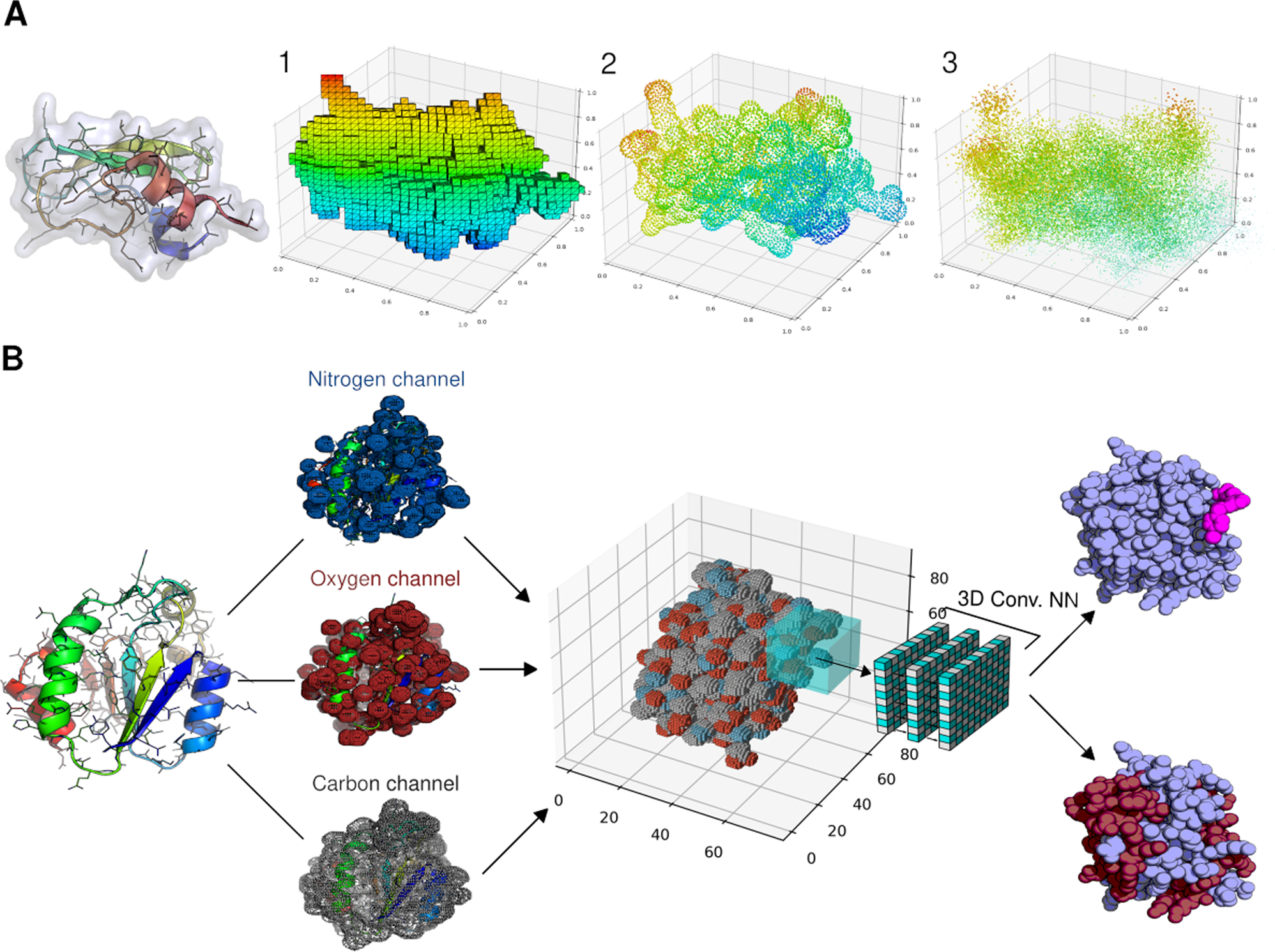
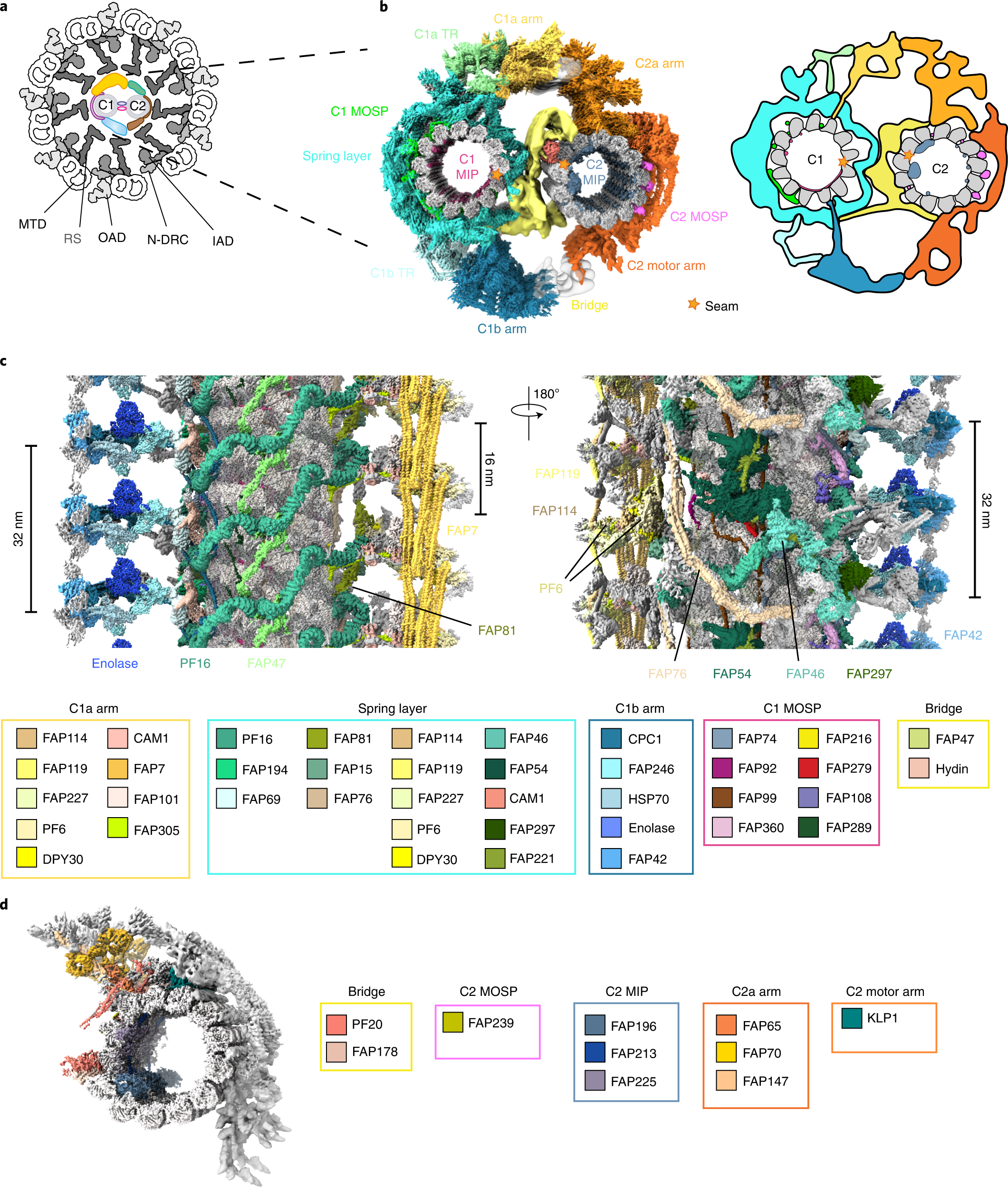
PyUUL provides an interface between biological structures and deep learning algorithms | Nature Communications
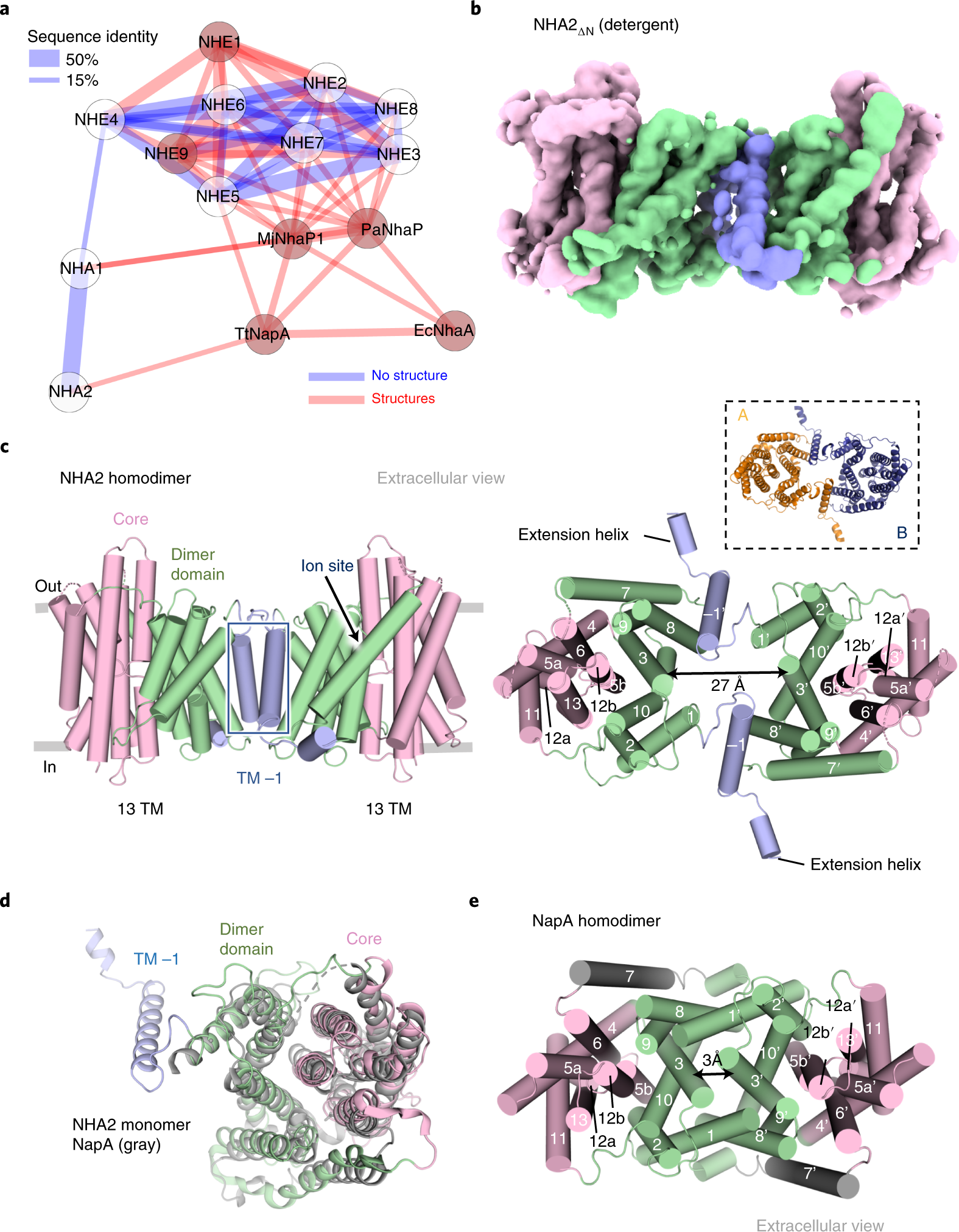
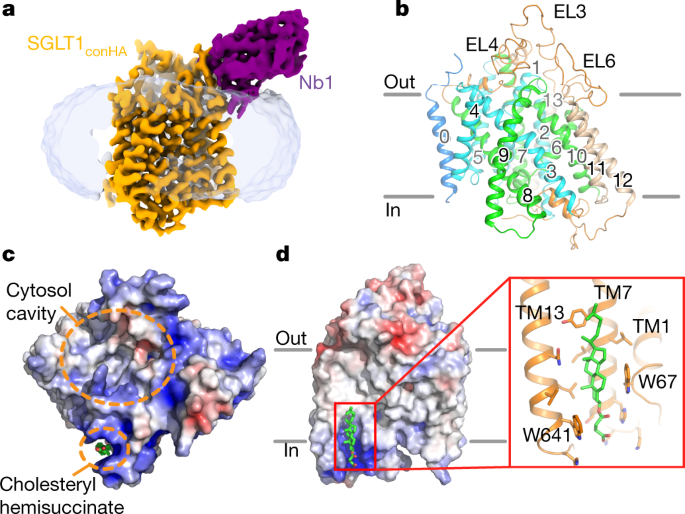
Structure, mechanism and lipid-mediated remodeling of the mammalian Na+/H+ exchanger NHA2 | Nature Structural & Molecular Biology
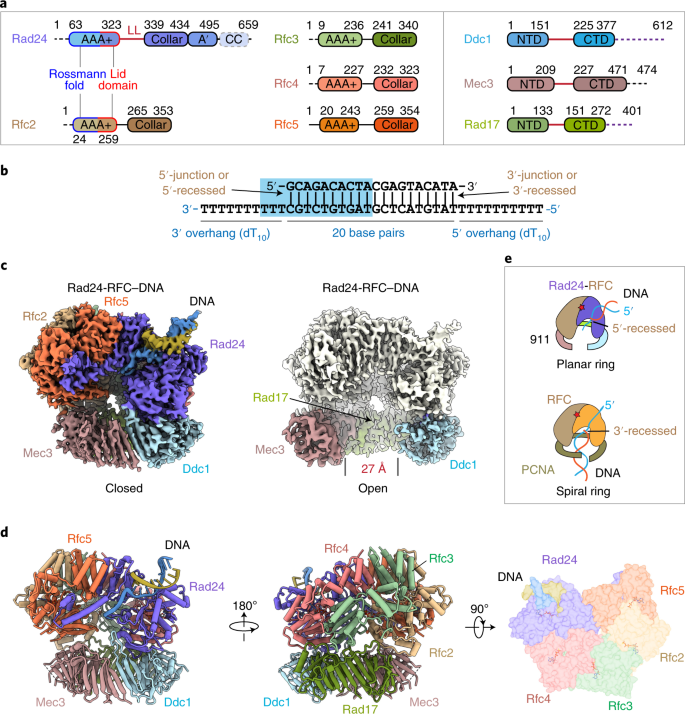
DNA is loaded through the 9-1-1 DNA checkpoint clamp in the opposite direction of the PCNA clamp | Nature Structural & Molecular Biology
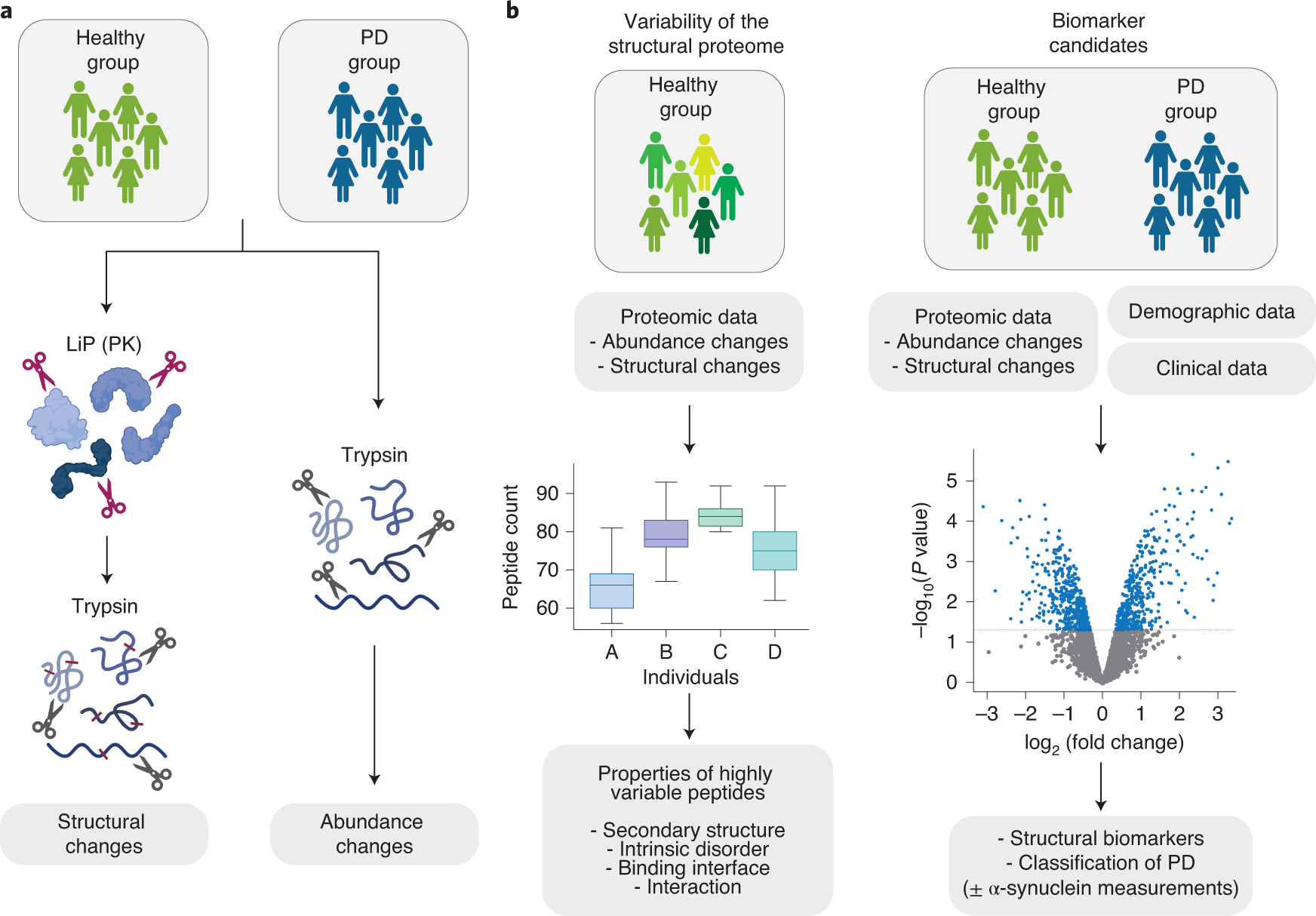
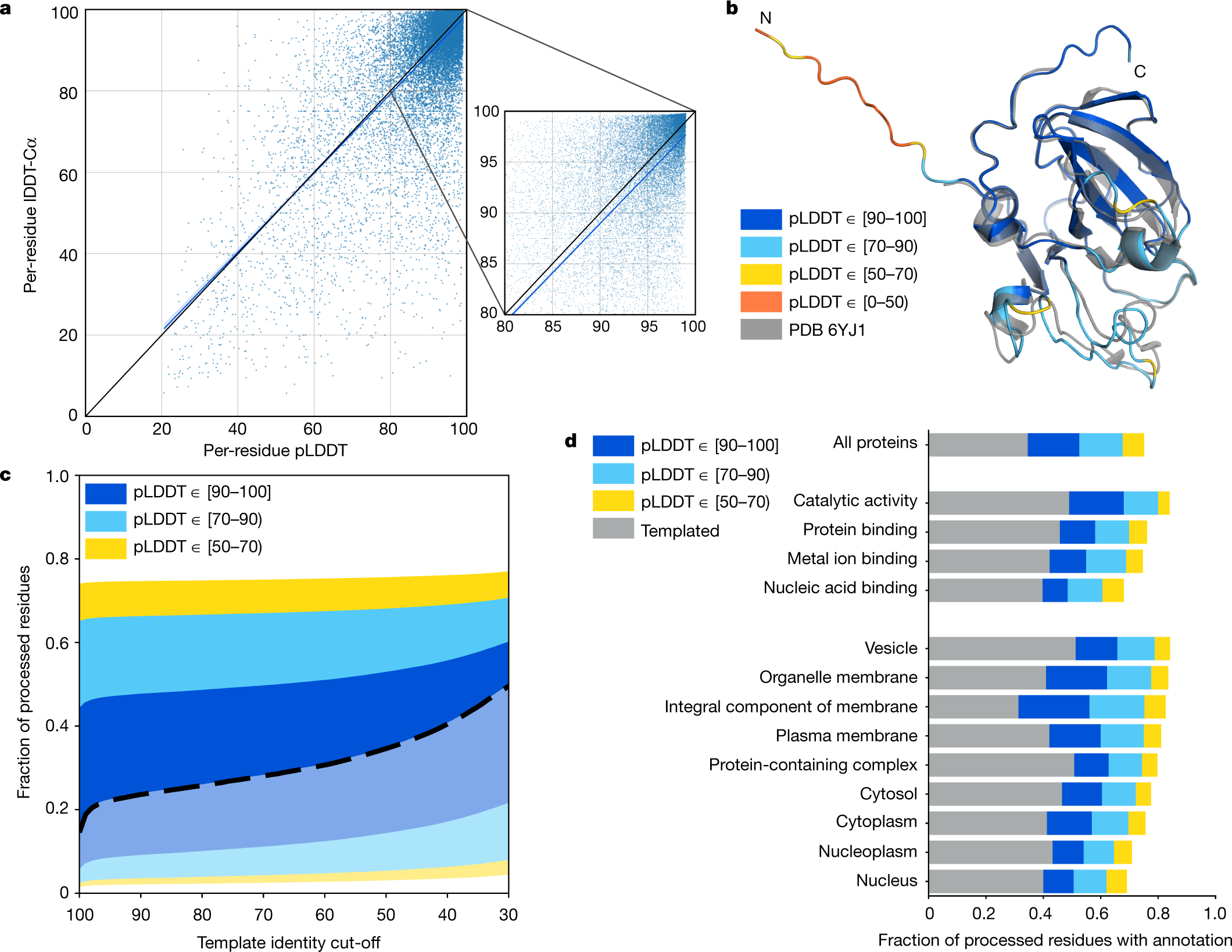
Global, in situ analysis of the structural proteome in individuals with Parkinson's disease to identify a new class of biomarker | Nature Structural & Molecular Biology

Cryogenic electron microscopy structures reveal how ATP and DNA binding in MutS coordinates sequential steps of DNA mismatch repair | Nature Structural & Molecular Biology

Conformational buffering underlies functional selection in intrinsically disordered protein regions | Nature Structural & Molecular Biology
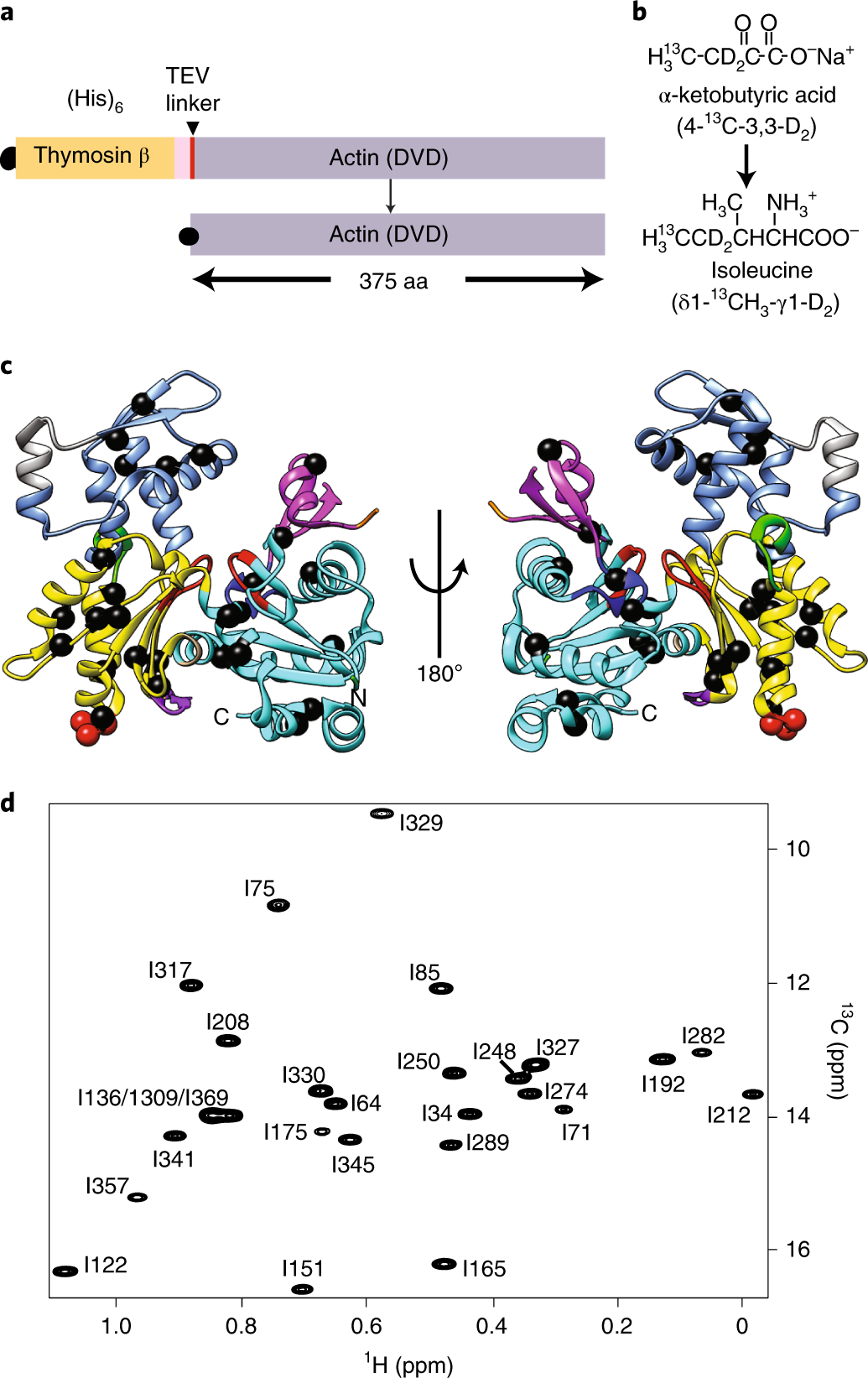
Bound nucleotide can control the dynamic architecture of monomeric actin | Nature Structural & Molecular Biology
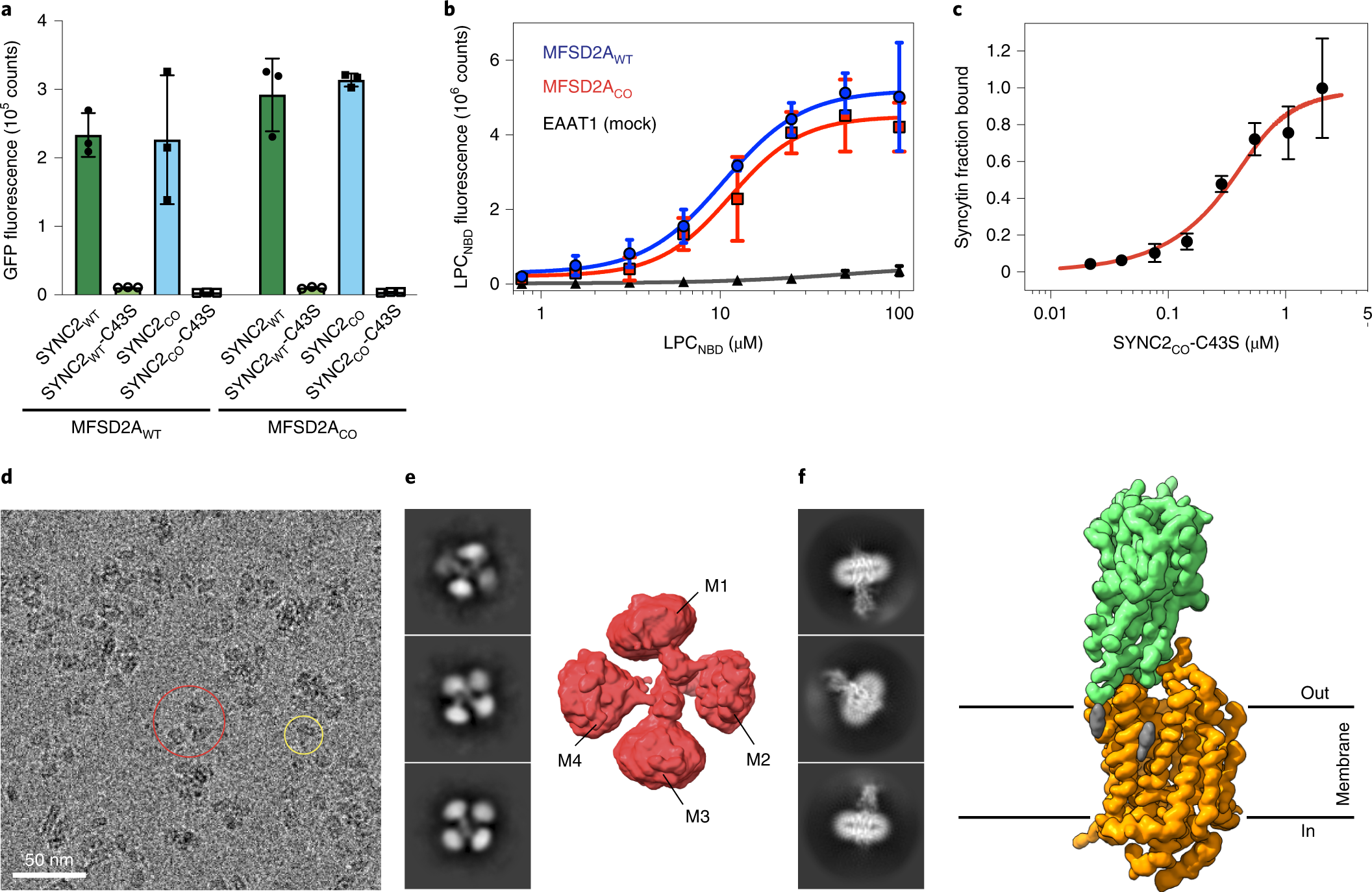
Structural insights into the lysophospholipid brain uptake mechanism and its inhibition by syncytin-2 | Nature Structural & Molecular Biology